Please put an active hyperlink to our site (www.rusnature.info) when you copy the materials from this page
Biomes and Regions of Northern Eurasia
Lake Baikal
<<< Geophysical Features of Lake Baikal and
its Catchment | Biomes & Regions Index | Aquatic Biodiversity >>>
Hydrology of Lake Baikal
Physical Characteristics
Physical limnology in Lake Baikal was pioneered by Vereschagin (1927, 1936, 1937), who
was first to establish that dependence of the temperature of maximum density (Tmd) on
pressure is of great importance for the pattern of water temperature (T) vertical
distribution. The water column of Baikal may be principally divided into two zones, based
on temperature profiles and water exchange processes: an upper zone, down to 250 m depth,
beneath which lies a lower zone, from 250 m down to the bottom sediments (Shimaraev, 1977;
Shimaraev and Granin 1991; Weiss et al, 1991).
Within the upper zone, Ņ passes through the state of Tmd twice a year, accompanied by:
(1) changes in temperature stratification (from inverted to direct in spring and vice
versa in late autumn); (2) changes in the coefficient of thermal expansion; and (3) the
development of free temperature convection. During these transitions, temperature
convection, wave action by strong winds and under-water currents ensure temperature
homogeneity of water within the active layer, and low stratification stability (otherwise
known as the state of homothermy). When summer and autumn stratification occurs, the
epilimnion and the thermo-cline are formed within the surface-water layer up to 25-50 m
depth, reaching annual maximum stability in August-September. During winter,
stratification becomes inverted, the thermocline within 25-100 m layer is eroded resulting
in a weaker stratification than in summer (Granin, 1991). In early summer, when spring
convection diminishes, the vertical Ņ profile in a 50-200 m water layer is close to the
Tmd profile; temperature decreases with depth due to increasing pressure (c. 0.21°C every
100 m) (Shimaraev and Granin, 1991). The lower boundary of the active layer (~220 m)
coincides with the occurrence of mesothermal temperature maximum (MTM) in winter, when Ņ
is equal to Tmd (c. 3.5°C). Its position is determined by the intensity of pre-winter
(November-December) water mixing and cooling within the active layer. In turn, the depth
of the MTM serves as a boundary for the development of spring convection (Vereschagin,
1936; Weiss et al, 1991; Shimaraev and Granin, 1991).
Within the deep-water zone, Ņ is greater than Tmd at all depths (Figure 17.3),
conditions that are typic for warm monomictic lakes (Vereschagin, 1936) and oceans
(Mamaev, 1987).
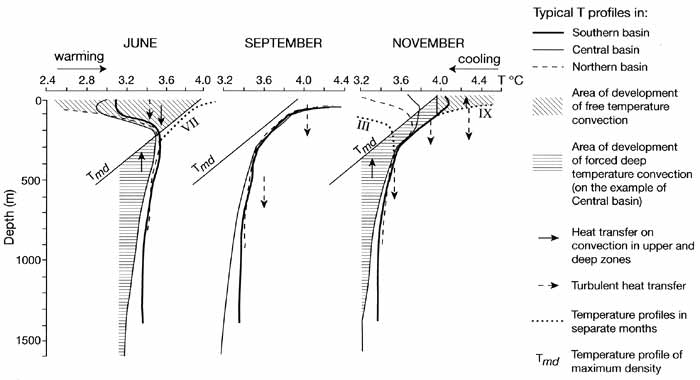
Fig. 17.3 Vertical distribution of convection in Lake Baikal
Vertical Ņ gradients are small (from 4-5 x l0-4 °C m-1 in 300-500 m layer
to 0.7-1.4 x 10 4 °C m-1 deeper than 1000 m) resulting in low stratification
stability, characterized by the Vaisala frequency close to the inertial frequency. Waters
in the central basin are more stratified and cooler than waters in the southern and the
northern basins (Shimaraev, 1977). For example, typical near-bottom temperatures within
each of the deepest depths of each of the three basins are about 3.30-3.35°C in the
southern, 3.10-3.15°C in the central, and 3.40-3.45°C in the northern basins. Deep-water
convection may be generated by forced sinking of colder, surface waters to 200-220 m
depths, which in turn are initiated by local and episodic atmospheric influences (Weiss et
al., 1991) or by water circulation on the front of spring thermobars (Shimaraev et al.,
1993).
Total dissolved solids (TDS) are low in open Baikal waters (about 96 mg I-1; Votintsev,
1993) and vertical differences do not exceed 1.5 per cent of TDS. Nevertheless, when Ņ is
close to 1mi and the coefficient of thermal expansion is near zero, the differences in TDS
may lead to water sinking within the MTM zone. This in turn causes thermobaric
instability, and results in deep-water convection. Cold water intrusion towards the bottom
of this zone is often accompanied by increases in ionic salinity (Hohmann etal., 1997),
thus testifying to thermohaline origin of deep-water convection. Spatial differences in
TDS may also contribute to ventilation and deep-water formation (Peeters et al., 1997;
Hohmann et al, 1997). For example, during the cold season, the Selenga River waters
containing up to 150 mg 1-1 of dissolved solids, sink along the Kukui canyon towards the
deepest depths in the central basin (Hohmann et al., 1997). On the other hand, water in
the northern basin is about 0.7 mg I-1 less mineralized compared to that in the central
basin, and this results in isopycnic water sinking from the central basin along the
Academician Ridge into the deep-water zone of the northern basin.
Deep waters are cooled in spring and early summer by convection, and are warmed in
autumn by heat transfer from the upper zone, resulting in seasonal temperature changes in
the order of 0.03-0.05°C. Turbulent heat flux into the deep zone is greatest in November
because of maximum wind activity at this time (Verbolov et al., 1965). Vertical transfer
of water within the field of horizontal currents may also be caused by deep-water renewal.
Sinking of surface waters occur in the coastal zone and also at the interfaces of
secondary circulation, whereas an ascent of deep waters takes place in the centre of the
lake (Verbolov and Shimaraev, 1973). Strengthening of general circulation results in
intensification of the near-bottom currents. In summer and winter the mean velocity of
currents between 20 m and 50 m is in the order of cm s-1, whereas within the
layer 50-100 m above the bottom, the velocity may reach 4-8 cm s-1. Within the
major part of the water column (between 200-300 m and the near-bottom layer), the velocity
is less than 2 cm s-1. During spring (May) and late autumn (October-November)
maximum currents at the near-bottom layer may be close to those observed in the surface
waters, up to 10-20 cm s-1 or more (Sokolnikov, 1960, 1964; Verbolov, 1996). As
a result of such circulation, the water above under-water slopes is significantly warmed
in summer and autumn. Therefore, the transition from direct to inverted temperature
stratification in the deep-water coastal regions advances later, in comparison to the
centre of the lake (Shimaraev et al, 1994).
Deep-water renewal may also be caused by processes which occur at thermal bars
(Shimaraev et al., 1993). Thermal bars arise in May, where warm riverine waters (T >
4°C) of large tributaries (e.g., the Selenga, Upper Angara, and Barguzin) come into
contact with colder lake water (T < 4°C). At these contact zones, denser (T = Tmd)
waters sink downwards, carrying away waters from both sides of the front (Figure 17.4).
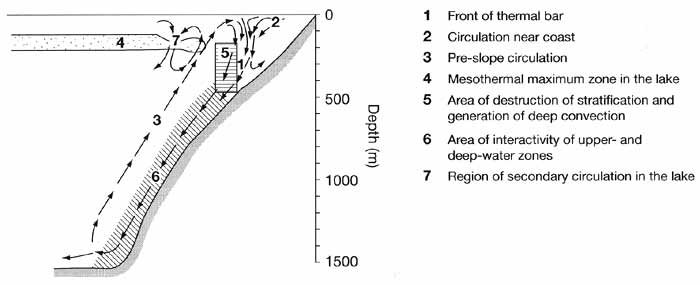
Fig. 17.4 Processes on the spring thermal bar near the eastern coast of
the central basin (according to Shimaraev et al., 1993, 1995)
Sinking of cold water (T = 3.1-3.4°C) initiates deep-water temperature convection
resulting in the near-front disturbance of winter stratification and complete homothermy
at depths greater than 150-200 m. Below the front, cold water moves downward along the
slopes. If the coastal water is more mineralized than open waters, sinking of these warmer
and denser waters occurs simultaneously. Both types of convection result in large-scale
circulation processes, with rapid (0.2-0.3 cm s-1) water sinking along the
underwater slopes, compensating for the ascent of deep waters directed towards the front
of the thermobar (Shimaraev et al., 1995). Deep-water renewal activity increases in the
coastal regions, resulting in oxygenation of deep waters (Shimaraev et al., 1996), the
input of plankton and suspended particles (Likhoshway et al., 1996), as well as an
increase in the number of microorganisms (Parfenova et al., 2000). This in turn stimulates
active internal cycling of organic matter in coastal regions of Baikal compared to the
open lake.
Estimated mean rates of vertical water exchange are based on determination of the water
age at different depths using dissolved atmospheric chlorofluorocarbons (CFCs) and 3H-3He
(Weiss et al, 1991; Peeters et al., 1997; Hohmann et al., 1998). Results obtained show
that in different parts of the lake the age of waters deeper than 250 m ranges from 7.2 up
to 11.1 years, corresponding to 9-13.8 per cent deep-water renewal. In 1992-95 the mean
rate of vertical water exchange through 250 m depth was 77.0 m a-1 in the
southern, 75.4 m a-1 in the central, and 59 m a-l in the northern basins,
respectively (Hohmann et al., 1998).
Water and Chemistry Budgets
The water budget is especially notable for the predomination of river influx (83 per
cent) over atmospheric precipitation (17 per cent) and the dominance of drainage from the
lake (81 per cent) over evaporation (19 per cent) (Shimaraev et al, 1994). Relatively few
tributaries contribute to the bulk of the input into Baikal, with the three largest (the
Selenga, Upper Angara, and Barguzin) contributing 47 per cent, 13 per cent, and 6 per cent
of the total annual river inflow, respectively. Only a further 12-15 other tributaries are
taken into account as 'gauged' rivers when estimating the total chemical budget of the
lake (Granina, 1997). Taking into account just the riverine input (since the atmospheric
input is close to evaporation), one can calculate from the data published by Afanasyev
(1976) and Gronskaya and litova (1991) that the entire water volume of Lake Baikal is
replaced every 377-400 years. This is termed the 'water residence time'.
About 98 per cent of TDS (in excess of 6 kilotonnes) enters the lake via rivers of
which approximately 14 per cent accumulates annually (Votintsev, 1961). Published data on
the chemistry budgets for the lake are discussed by Granina (1997). Until recently,
particulates have not been included in existing budget estimates, even though they
comprise up to 30 per cent of the total input of chemicals into the lake. Since 1993,
however, data on the composition of suspended particles in rivers have been obtained,
enabling for the first time complete budgets (both dissolved and particulate) to be
constructed for some elements (Callender and Granina, 1995, 1997a, b). These budgets are
crucial in determining different external and interlacustrine processes. For example,
biogeochemical silica mass balance (Figure 17.5) shows that the production of diatoms
(siliceous microalgae) in the lake is supported mostly by remineralization processes in
the water column but is accompanied by a significant contribution from riverine inputs of
dissolved silica.
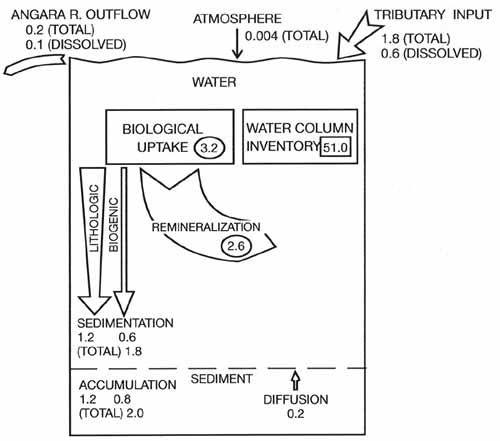
Fig. 17.5 Box diagram of silica mass balance and internal cycling in
Lake Baikal.
Fluxes are expressed as million tonnes of silica per annum and water column inventory as
million tonnes of silica.
After Callender and Granina (1995)
Chemical Constituents of Lake Water
Comprehensive water chemistry datasets were collated in the 1960s by Votintsev (1961)
and Votintsev et al. (1965), and have since been updated by Falkner et al. (1991) (Table
17.2). The pH of the lake water is generally 7.1-7.2, although photosynthetic activity may
result in a larger range (7.1-8.6) (Votintsev, 1961; Falkner et al., 1991). The total
mineral content of the lake water is relatively low (94-96 mg I-1), reflecting the
chemistry of the tributaries, which is controlled predominantly by the weathering of
igneous crystalline rocks (typical in the lake drainage basin). Generally, the major ion
composition is dominated by Ca2+ and HCO3; most of the tributaries have similar major
ionic compositions (Votintsev, 1961).

Table 17.2 Concentration of major ions in Lake Baikal water (ppm)
In accordance with Votintsev (1961) and recent data published in Falkner et al (1991;
1997), chemical constituents in the lake can be divided into three broad groups:
- 1. Major ions and some minor elements, whose distribution is characterized by a uniform
pattern of vertical distribution and by an absence of seasonal changes (except within the
regions under tributary influence).
- 2. The components whose concentrations increase with increasing water depth: nitrate
nitrogen (N-N03), phosphate phosphorus (P-P04), silicon (Si), germanium (Ge), iron (Fe),
and manganese (Mn).2
- 3. The constituents whose concentrations decrease with water depth: oxygen (02), organic
matter (OM), and barium (Ba).
Residence times for many of the major ions (Ca2+, and SO24) are similar to the
estimated residence time for water (Falkner et al., 1997), indicating that these ions are
in steady state with respect to their riverine inputs. This refutes claims by other
authors that ions, such as sulphate, are increasing due to anthropogenic activity (e.g.,
Tarasova and Mescheryakova, 1992). Concentrations of all the other non-major ions are
controlled by biological and hydrological processes in the water column. For example,
nitrate and phosphate concentrations show seasonal changes in the trophogenic layer, with
minima occurring every spring and summer due to increasing phytoplankton numbers
(Votintsev, 1961; Tarasova and Mescheryakova, 1992). These seasonal changes tend to
deplete nutrients up to 100 m depth, during which time nitrate may limit productivity as
it becomes severely depleted (Kozhov, 1963; Weiss et al., 1991), while phosphate levels
rarely fall to limiting levels (Weiss et al., 1991; Tarasova and Mescheryakova, 1992).
Concentrations of silicon are 3.5 times lower than that in riverine waters because of
intensive silicon uptake by diatoms that predominate in the plankton community (Votintsev,
1961). Vertical profiles of silicon show surface-water depletion and deep-water enrichment
due to silicon uptake by diatoms in surface waters and its release by dissolution of
valves sinking into deeper waters (Votintsev, 1961; Votintsev et al., 1965; Falkner et
al., 1997). Silicon enrichment of deep waters is also caused directly by riverine input,
especially in the central basin (Domysheva et al., 1998). The depth-weighted average of
silicon concentration in each of the three basins is 0.85 mg H (south), 1.06 mg I-1
(central), and 0.95 mg I-1 (north) (Domysheva et al., 1998). Consistent with the known
accumulation of biogenic silica in Baikal sediments, the residence time of silicon is c.
170 yr, significantly less than the water residence time of the lake (Falkner et al.,
1997).
Oxygen concentrations in the upper water layers range from 94 per cent to 104 per cent
saturation, whereas saturation of near-bottom waters is usually less than 80 per cent
(Votintsev, 1961; Weiss et al. 1991; Shimaraev et al., 1996). The high oxygen saturation
of surface waters reflects the combined effects of atmospheric exchange, photosynthetic
production in the photic zone, and respirative consumption (Weiss et al., 1991). Oxygen
consumption within the deep-water zones of the central and northern basins ranges from 1.1
to 3.4 umol I-1a-1 (Shimaraev et al., 1996). An attempt to estimate the
atmospheric oxygen exchange exhibits substantial spatial differences across the lake; the
average oxygen uptake is much higher during the long period of autumn-winter cooling
compared to spring warming (Mizandrontsev et al, 1996, 1998).
Trace elements have been poorly investigated in Lake Baikal because of their extremely
low concentrations, which approach general detection limits; this is changing with better
practice and state-of-the-art techniques (e.g., Falkner et al, 1991, 1997; Grosheva et
al., 1993). The vertical distribution of trace elements in Lake Baikal is rather uniform
(Vetrov and Kuznetsova, 1997), although there are some notable exceptions. For example,
the vertical profile of iron shows a sharp gradient within the photic zone, possibly
caused by photosynthetic activity (Leibovich-Granina, 1985); the same may be a case for
vanadium, nickel, and copper (Falkner et al, 1997), whilst both iron and manganese
concentrations increase in the near-bottom layer (Leibovich-Granina, 1985).
Atmospheric fluxes of elements entering the lake are low (on average about 3 per cent
of the total input) although exceptions do exist for some industrial pollutants
transported by air, such as zinc, copper, chromium, and in particular, lead (Boyle et al.,
1998; Granina et al., 2000). Atmospheric fluxes of these trace elements can reach 25 per
cent, and thus pose a significant threat of pollution to the lake. Furthermore,
atmospheric fluxes on the lake surface are higher when one takes into account the input of
elements with aerosol particles (Khodzer et al., 2000). It should be noted here though,
that overall, modern riverine fluxes of major ions and nutrients are higher compared to
those estimated 20-40 years ago (Sorokovikova et al., 2000). It is clear therefore, that a
new approach to construction of a more accurate chemical budget is still required.
<<< Geophysical Features of Lake Baikal and
its Catchment | Biomes & Regions Index | Aquatic Biodiversity >>>
Contents of the Lake Baikal section:
Other sections of Biomes & Regions:
|
|